Every second of every day your body relies on soluble major and trace mineral ions to conduct and generate billions of tiny electrical impulses. Without these impulses, not a single muscle, including your heart, would be able to function.
Minerals and The Calcium Myth
First, let’s talk about calcium. We live in a calcium obsessed society. Every health expert tells us to supplement with more and more calcium. Right now, coral calcium is one of the hottest supplements in the alternative health market. But the simple truth is: the vast majority of people get more than enough calcium in their diets. In fact, most people get too much. The average American consumes 600 lbs of dairy products a year. That’s almost 2 lbs per day, and yet osteoporosis rates in the United States are among the highest in the world. In fact, not long ago, researchers at Yale University analyzed 34 published studies from 16 different countries and found that the countries with the highest rates of osteoporosis (including the United States, Sweden, and Finland) were those in which people consumed the most meat, milk, and other animal-based foods. The study also showed that African-Americans, who consume, on average, more than 1,000 mg of calcium per day, are nine times more likely to experience hip fractures than are South African blacks, whose daily calcium intake is only about 196 mg. On a nation-by-nation basis, people who consume the most calcium have the weakest bones and the highest rates of osteoporosis.
The interesting thing is that our world-wide obsession with calcium actually started in the 1950’s under pressure from the American dairy industry. Before then, historically, people didn’t consume much calcium – and had very few problems with osteoporosis. What they did consume was magnesium – almost 5 times as much magnesium as we consume today. Which brings up a key point: the health of our bones depends far more on other factors such as magnesium, boron, and vitamin D than it does on calcium. In fact, consumption of too much calcium is just plain damaging to your bones and your health in general.
Ah, but what about pH? Isn’t calcium good for raising pH? All of the ads for coral calcium say that calcium is great for raising pH, and isn’t that important? Absolutely! But calcium isn’t the only way to raise pH; there are many other ways to raise it – including consuming less dairy, meat, and soda pop, for example. And by consuming more magnesium.
Magnesium
Without calcium, you cannot live, but without adequate amounts of magnesium to balance that calcium, you will find yourself falling victim to hardening of the arteries, arthritis, diabetes, and senility. Magnesium is the activating mineral for close to 400 different enzyme reactions in the body (that we know about) – more than any other mineral. Too little magnesium literally impacts your body negatively in hundreds of ways. And what makes the problem even worse is that magnesium is much harder for your body to absorb and utilize than calcium. This fact alone makes a joke of the standard 2:1 ratio of calcium to magnesium found in most supplements. Based on absorption, the ratio provided by most supplements is much closer to 6:1 or even 8:1 in favor or calcium — a very unhealthy ratio. The bottom line is that magnesium is the most important major mineral needed by your body, and unfortunately, the one that is most often depleted.
Trace Minerals
Ten years ago, few people knew much about trace minerals. They were almost never included in any commercial vitamin/mineral formula. But today, almost everyone knows that trace minerals are, quite simply, those elements needed by your body in very small amounts (less than 100 mg a day) for health and life. Every second of every day your body relies on soluble major and trace mineral ions to conduct and generate billions of tiny electrical impulses. Without these impulses, not a single muscle, including your heart, would be able to function. For that matter, your brain would not function, and cells would not be able to use osmosis to balance water pressure and absorb nutrients. Among other things, soluble minerals are:
- Ionized conductors of the body’s electrical current necessary for body functions.
- Catalysts and activators of other nutrients.
- Building blocks of enzymes, hormones and other natural agents used by the body to perform specific functions.
- Equalizing and balancing. Body fluids, fluid pressures, and pH are balanced with minerals. Also, it is known that in the absence of certain trace minerals, certain heavy metals are more likely to accumulate. Trace minerals are often administered in hospitals as part of the treatment for such disorders as lead poisoning.
- Even for digestion and assimilation, minerals are essential. High amounts of chlorides, for example, are necessary for the body to make hydrochloric acid. Still other minerals are used in the assimilation process.
Traditionally, eating fresh grains, fruits, and vegetables grown in nutrient-rich soil and drinking mineral rich water have been the primary supply for the full spectrum of ionically charged minerals. Unfortunately, naturally occurring, nutrient-rich soil is almost non-existent on commercial farms. Aggressive farming techniques have stripped most trace minerals from the soil. The simple truth is that the use of nitrogen fertilizers to make plants grow is relatively cheap, and is therefore the foundation of modern farming methods. Remineralization of the soil, on the other hand, is very expensive, and produces no “visible” result. It merely makes the food healthier to eat…but that of course has no value when sitting in the produce section of a supermarket.
On the other hand, when people consume a diet derived from such depleted crops, the intake of essential trace minerals becomes inadequate. This leads to poor health and disease. What are some of these key trace minerals? Actually, there are several dozen of them, including copper, tin, silver, gold, and lithium, etc; but three that have recently been much in the news are:
- Selenium, essential for preventing cancer
- Boron, essential for preventing osteoporosis
- Chromium, essential for regulating blood sugar levels
Minerals From the Sea
Each of us carries in our veins a salty river in which the elements are combined in almost the exact same proportions as in sea water. This unique combination of elements is found not only in our bodies, but also in the bodies of virtually all living creatures. Even the protoplasm that streams within each cell of our bodies has this same chemical structure impressed on it.
This same equilibrium can also be found in the seas around the world where minerals and trace minerals have collected and concentrated in liquid ionic form since the creation of the earth itself. All the seas of the earth contain basically the same balance of minerals including Utah’s Great Salt Lake. In fact, 84 of the earth’s basic elements have been identified in sea water (44 are considered to be the most common soluble minerals). It is astounding to realize that the dynamic equilibrium that takes place with liquid ionic minerals and trace minerals has created the same basic balance in sea water that is found in healthy blood plasma and lymphatic fluid.
The bottom line is that the best, most balanced, concentrated source of essential elements for our bodies is sea water. And the purest, most concentrated sea water in the world comes from the Great Salt Lake. (Note: 99.5% of the sodium has been removed)
| Decreasing Average Concentration of 66 Elements in Great Salt Lake Water (mg/l) |
|||||
|---|---|---|---|---|---|
| Oxygen | Boron | Iron | Vanadium | Selenium | Gold |
| Hydrogen | Silicon | Indium | Titanium | Germanium | Niobium |
| Chlorine | Fluorine | Molybdenum | Cesium | Xenon | Thallium |
| Sodium | Argon | Zinc | Cerium | Chromium | Hafnium |
| Magnesium | Nitrogen | Nickel | Antimony | Thorium | Helium |
| Sulfur | Lithium | Arsenic | Silver | Gallium | Selenium |
| Calcium | Rubidium | Copper | Yttrium | Mercury | Tantalum |
| Potassium | Phosphorus | Tin | Cobalt | Lead | Beryllium |
| Bromine | Iodine | Uranium | Neon | Zirconium | Protactinium |
| Carbon | Barium | Krypton | Cadmium | Bismuth | Radium |
| Strontium | Aluminum | Manganese | Tungsten | Lanthanum | Radon |
Less Toxins
It is important to understand that virtually all food contains small amounts of toxic elements. Just as good elements are pulled out of the soil and become part of the plant, so too the bad elements. There is no avoiding it. The key is that, in most cases, they are in such small amounts in our food that they do not affect our health negatively. It only becomes a problem when considered cumulatively. That is: you get the toxic elements in small amounts in your food and in your water and in the air you breathe and in your toothpaste and in your amalgam fillings etc. The bottom line is that any way you can reduce your exposure will have long term benefits for your health. The simple fact is that the percentage of toxic elements in Great Salt Lake minerals is extremely low.
Mineral Absorption
Probably nothing is more confusing to people than the arguments that wage back and forth as to what form of minerals your body can best utilize. Keep in mind that you cannot benefit from minerals unless two things happen
- You absorb the minerals in your intestinal tract
- Your cells then utilize those absorbed minerals
Minerals are absorbed via active and passive transport. Active absorption (via ionically charged minerals) is more efficient, but is also limited since different nutrients compete for the same charged receptor sites. Passive absorption (via food bound minerals) is less efficient, but is not limited by the receptor sites. Most minerals are absorbed at a rate of 6% to 40%, except for sodium and potassium, which are absorbed at about 90%. However, absorption rate and bioavailability depend on several factors, including pH, the source of the minerals, physiology, and the presence of other minerals.
- Minerals are charged elements, so they are better absorbed in an acidic environment. As people age, they tend to produce less stomach acid, which means they also tend to absorb fewer minerals. (Not to mention the use of acid inhibiting drugs now being mass marketed that further impair mineral absorption.)
- The food source can either help or hinder mineral absorption. For example, the iron found in meat is two to three times more absorbable than plant based iron salts.
- Plants contain compounds, including phytates, fiber and oxalates that lower mineral absorption, particularly calcium, iron, magnesium and zinc. Phytates found in unfermented soy products and unprocessed whole-grain products bind minerals and lower absorption. Oxalates found in spinach and rhubarb can reduce calcium absorption — in some cases to as little as 3% to 5%.
In most cases, ionically charged non-food-bound minerals, which are actively absorbed, are more absorbable than food-bound minerals since they avoid many of the binding problems associated with food. Another advantage they have is that they do not require stomach acid to acquire their charge, as they come “pre-charged.” As mentioned above, this is particularly important as people get older or use over-the-counter acid reflux medicines since their stomachs no longer produce enough acid to “ionize” the minerals found in the food they eat. Using a low-sodium, ionic-trace-mineral supplement avoids that problem.
The lowest absorption, however, is reserved for a third form of minerals (non-ionic, non-food-bound minerals), which provides the worst of all possible worlds and offers absorption rates of 3%-5% across the board, while still limiting the amount that you can absorb through the charged receptor sites. Unfortunately, this is the form most frequently used in mass-marketed supplements. Why? Because it’s cheap.
Recognizing the absorption problem, many supplement makers look to overcome the absorption problem associated with these type of minerals by chelating them, a process that mimics the food binding found in nature and that allows the mineral to take advantage of the excellent absorption abilities of amino acids – to, in effect, use the amino acids as a transport system and cross the intestinal lining more easily. Unfortunately, chelated minerals are neither “fish nor fowl.” And even though you can fool the intestine into absorbing them, the cells of your body tend not to like them.
Bottom line: the most absorbable form of mineral is the ionic mineral. The reason is quite simple. The unstable ionic state allows the mineral to bond readily with water, making it possible for the body to easily absorb it. (Note: the type and structure of the water used plays a key role in regulating absorption, which we will talk about in detail shortly.) Minerals that are absorbed in their ionic form are in true liquid solution and have either positive or negative charges. They also have unique properties that distinguish them from each other and allow them to freely take part in biochemical communication throughout the body. These communications help nutrients move to those areas of the body where they are most needed. Which then brings up the question, “What happens when the mineral arrives at the cell where it’s needed?”
Getting Minerals Into the Cells
Absorption in the intestinal tract only gets you half way to utilization. Once you get past the intestinal tract, you still have to get the trace minerals into the cells where they actually do some good.
It is here that the water used to put the minerals into solution plays a major role. And to explain what happens here, let’s look at your laundry.
Although all water consists of the same basic H20 molecules, water nevertheless varies according to how these molecules bond together to form “water molecule groups.” To put it simply, it is in the size of these groupings that water differs. Ordinary water has electric charges that cause it to clump together. We see this as surface tension that causes water to bead up on the surface of a car. At the atomic level it causes the water molecules, which are normally arranged in tetrahedral structures, as seen below,
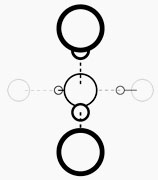

to clump together in large groupings containing many tetrahedrons stuck together. In the same way that these large groupings of many tetrahedrons cannot easily pass in and out of the tightly bunched fibers of your clothing, they cannot easily pass through the walls of living cells. They are not bio-available.
What laundry detergents do is use chemical surfactants to help break apart these large water molecule groups into smaller groups. This, in effect, makes the water “wetter” – better able to penetrate clothing fabric and fibers, and thus, wash away dirt. The same principle holds true with the water we drink. If the natural surface tension of the water is causing it to cluster in large molecule groups, it cannot easily enter or leave cells – making it an inefficient transporter of nutrients into the cells…and waste out. If we can find a way to break the water into smaller clusters (obviously without laundry detergent chemicals), we can then make the water more efficient in its task.
There are several ways to accomplish this (magnetic fields, sound waves, etc.), but one of the best is to use a micelle catalyst. The advantage of the micelle is that it not only causes the tetrahedral clusters to separate, it actually causes the tetrahedrons themselves to break apart and reform into “chain” structures. To put it another way, the best water to use, the water that most enhances the bio-availability of the minerals in question is CAW (Catalyst Altered Water). In effect, CAW has been electrically altered by the addition of the micelle catalyst so that it no longer clumps together. The rigid tetrahedral structure of the water breaks apart so that the water flows much more easily in and out of cells – readily carrying minerals and nutrients into the cells, and waste out. Understand, CAW is not a nutrient, but a vehicle by which nutrients are carried throughout the body’s cells, and by which waste is carried away from the cells – with water as the means of transportation. Water which has been altered by the micelle catalyst becomes “wetter” and more efficient. So what is micelle?
Micelle is an extremely small, high-energy particle with a powerful negative magnetic field. As already mentioned, when added to ordinary water, it causes a change in the structure of that water. The molecular structure is altered from the very stable tetrahedron structure into chains of water molecules attracted to the colloidal micelle by strong electrostatic bonds.
A simple way of explaining this is say that CAW is a more efficient form of water. When consumed regularly, CAW can help in the absorption and assimilation of all nutrients (not just minerals), increase enzyme activity, and strengthen the immune system. And in addition to all of those benefits, regular consumption of CAW offers increased elimination of toxins and wastes. It also works as an extraordinary free radical scavenger and helps reduce inflammation — throughout the body.
CAW is very concentrated. A mere one ounce will optimize a full gallon of regular water.
Catalyst Altered Trace Minerals™ Alkalinize Your Body
One of the major benefits of a low-sodium catalyst-altered trace mineral formula is that it can balance pH – raising low pH and lowering high pH. As detailed in Lessons from the Miracle Doctors, maintaining a blood pH of around 7.45 is vital. Any extended pH imbalance of any kind threatens our well being — threatens, in fact, our very lives. Managing the pH balance of all of our bodily fluids is so important that our bodies have developed systems to monitor and balance acid-alkaline levels in every cell and bio-system.
Our bodies function in a very narrow range of acid/alkaline balance (pH). Our blood in particular is very sensitive to these changes. Ideally, blood pH should be slightly alkaline at about 7.45. If it varies by even as little as a few tenths of a point, severe illness and death may result. Unfortunately, most of the food we eat is highly acidic (meat, dairy, sodas, alcohol, cooked grains, etc). In the end, it becomes too much for our bodies to handle. If we don’t correct the problem by “alkalinizing” the body, disease, sickness, and death are the inevitable result. Fortunately, regular consumption of Catalyst Altered Trace Minerals™ can help in this regard. (It is important to note that Catalyst Altered Trace Minerals™ do not raise the pH of the water they are added to. They do, however, raise the pH of the body itself by neutralizing excess acid in body fluids and tissues, converting that acid into salts, and then passing those salts out of the body in the urine.)
Benefits of Catalyst Altered Trace Minerals™
By blending ionic trace minerals from the Great Salt Lake with catalyst altered water, you end up with an extremely high-powered, nutrient-dense concentrate — one 16 ounce bottle transforms a full 16 gallons of regular drinking water — with the following benefits.
- It provides a full complement of the trace minerals your body needs.
- It produces a more bio-active form of water.
- It increases the bio-magnetism of living cells up to 50 percent.
- It works as a powerful antioxidant.
- It makes water more “refreshing” and “hydrating” than uncatalyzed water.
- It is highly concentrated. A few drops will transform an entire glass of water. A mere ½ ounce will optimize a full 8 glasses (64 ounces) of water – an entire day’s supply for most people.
- It is high in magnesium (300 mg per day), which is even more important for most people than calcium.
- It improves bowel activity because of the high level of magnesium.
- It is high in chloride (816 mg), the principle negatively charged ion in the body that serves as one of the body’s main electrolytes.
- It is very clean, much lower in potentially “toxic” elements than any other source of trace minerals.
- It is easily absorbed – both because the minerals are in an ionic form and because the CAW helps the minerals pass easily through cell walls.
- It balances out body pH.
- It improves the efficiency of all the body’s biological systems.
Using Catalyst Altered Trace Minerals™
Since the benefits of using catalyst altered trace minerals goes far beyond the minerals themselves, into the very ability of the concentrate to change the structure of all the water we drink, it is best to add them to all of the water or juice you consume during the day. Fortunately, the concentrate actually improves the taste of water, giving it a slightly sweet, mineralized, mountain-stream kind of flavor. One ounce of concentrate will catalyze an entire gallon of water. That means that you only need 1/2 ounce per day to optimize and remineralize 8 glasses of water, the amount most people should drink each day. (Or you can just add 24 drops per 8 ounce glass of water.) One 16-ounce bottle of concentrate will transform 16 gallons of regular drinking water. That’s a 32-day supply at 8 glasses per day.
Still interested in more information on this topic? Review the following Newsletters:
—
This product is no longer available by Baseline Nutritionals.












Were can I purchase catalyst
Were can I purchase catalyst altered trace minerals (CAW)?
You can find all Jon Barron's
You can find all Jon Barron's recommended formulas on the Products page above, and a list of companies that sell them.
Catalyst Altered Trace
Catalyst Altered Trace Minerals – where can I find it.
Also, I have a whole house water filter by Filtercon, similar to Water Boy. Will filter remove any Pharmaceuticals. It tasts like my sparklettes bottle water, I live in San Diego county, CA
Should the minerals be added to distilled ater or my filtered water.
Thanks,
Don Woolery
Hi Don,
Jon Barron always
Hi Don,
Jon Barron always recommends to add minerals to water, especially distilled water. To find Jon's recommended products, go to our products page above and look for "Liquid Trace Minerals," fourth one down the chart. On the right of the chart is the list of companies who sell them.
Question. Can I add the
Question. Can I add the product to hot liquid specifically to the green tea
Thanks
It certainly won’t harm the
It certainly won’t harm the minerals, if you add them AFTER boiling or steeping, but I wouldn’t bet on the taste if you add it to tea. No serious tea drinker would want the odd taste in their tea.
Can I add the product to a
Can I add the product to a smoothie?
Of course, smoothies, water,
Of course, smoothies, water, juice, pretty much anything works.
What exactly is the "micelle
What exactly is the "micelle catalyst" used in this product, which my wife purchased? This article is very vague on the topic. Is it a chemical substance? Is it an extract of some natural product? I would think you would have to list this as an ingredient. Unless of course it's an "energy" product. I would like to know what my family is taking if they consume this product. Perhaps others might be curious too.
Micelle is a term used in
Micelle is a term used in chemistry. It is defined as an electrically charged particle formed by an aggregate of molecules and occurring in certain colloidal electrolyte solutions. The one used in Catalyst Altered Trace minerals is naturally occurring.
Thank you. What is the name
Thank you. What is the name of the micelle used in Catalyst Altered Trace Minerals, or the chemical compound?
The micelle catalyst used in
The micelle catalyst used in Catalyst Altered Trace Minerals is created by combining four substances found in nature using a complex, patented process. It is highly diluted when mixed with water — about 1 part to a 1,000. In this case, diluted actually works better than concentrated. More than that, we cannot say as we are starting to cross over into formulary trade secrets.
Why is this product, Catalyst
Why is this product, Catalyst Altered Trace Minerals, no longer offered please? And do you recommend an alternative product?
If you go to the product page
If you go to the product page, we recommend this company now: http://www.traceminerals.com/products/liquid-tablet-minerals/concentrace-ionic-minerals
I clicked product on your
I clicked product on your menu
to order some products but I don’t see any link for order.
Would you tell me how.
Thanks
Gil
Yes, they no longer sell this
Yes, they no longer sell this formula. If you go to the product page, we recommend this company now: http://www.traceminerals.com/products/liquid-tablet-minerals/concentrace-ionic-minerals