We now explore how what we’ve learned about the anatomy and physiology of the stomach translates into problems that can occur, including peptic ulcers, acid reflux, mineral absorption, B12 absorption, incomplete digestion, and weight gain – and how to deal with those problems.
 In the last newsletter (part 3 in our series on the digestive system), we concluded our discussion of the anatomy and physiology of the stomach from a natural health perspective. In this issue, we take the logical next step and explore in detail the things that can go wrong with your stomach. Amusingly, the common stomach ache is not one of them. When most people complain of a stomach ache, they put their hands over their transverse colons — the source of the problem — and an area we will cover in great detail later in our series on the digestive system. But for now, our focus will be on stomach/duodenal specific problems. These include:
In the last newsletter (part 3 in our series on the digestive system), we concluded our discussion of the anatomy and physiology of the stomach from a natural health perspective. In this issue, we take the logical next step and explore in detail the things that can go wrong with your stomach. Amusingly, the common stomach ache is not one of them. When most people complain of a stomach ache, they put their hands over their transverse colons — the source of the problem — and an area we will cover in great detail later in our series on the digestive system. But for now, our focus will be on stomach/duodenal specific problems. These include:
- Peptic ulcers
- Acid reflux
- Mineral absorption
- B12 absorption
- Incomplete digestion
- Satiety and weight gain
- Hiatal hernias would also be an issue, but we covered those two issues ago)
So, without further ado…
Peptic ulcers
Most people do not understand ulcers. They think they are either caused by too much stomach acid (not true) or caused by the bacteria H. pylori (only partially true). They also think most ulcers occur in the stomach (gastric ulcers), which again is not true. In fact, about 60% of all peptic ulcers occur in the duodenum, where stomach acid is actually neutralized shortly after making its appearance — which surprisingly contributes to the problem.
Quite simply, a peptic ulcer is any ulceration in acid-exposed areas in the duodenum or stomach. Stomach acid itself is not the culprit here. After all, strong stomach acid is a normal part of digestion. In fact the key to understanding peptic ulcers lies in two words found in the definition above: “acid-exposed.” The bottom line is that peptic ulcers occur when the mucous that lines every square inch of the stomach and duodenum and that protects them from the corrosive effects of stomach acid is somehow worn away from an area of tissue — exposing that tissue to the burning effects of the acid. Peptic ulcers, then, are caused not by stomach acid, but by damage to the body’s protective mucosal lining.
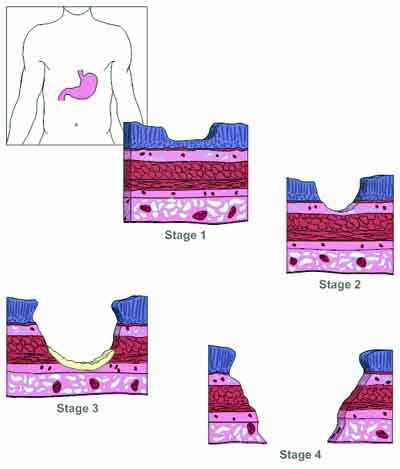
This can have several causes:
Helicobacter pylori (H. pylori) is considered the primary culprit. Although somewhat resistant to stomach acid, H. pylori bacteria cannot really withstand a full onslaught of undiluted acid. Therefore, it lives under the mucosal layer lining the stomach, but does not actually invade it. It thus protects itself from the gastric juices, which can destroy it. H. pylori further protects itself by secreting urease, an enzyme that breaks down urea into ammonia and carbon dioxide; the ammonia in turn neutralizes stomach acid. This helps it survive short bursts of exposure to less than full strength stomach acid as it makes its way through the stomach and on into the duodenum. As the organism thrives and expands its colony under the mucosal lining, it causes the lining to inflame. This causes a thinning and breakdown of the mucous layer that protects the lining. The lining of the duodenum or stomach is now exposed to acid and pepsin, and ulcers may develop.
So again, stomach acid does not directly cause the stomach ulcer, and — here’s an important point — can actually kill the bacteria if it is strong enough and is present before the bacteria can establish itself under the mucosa. If stomach acid is diminished for any reason (such as regular use of proton pump inhibitors, excessive use of antacids, or regular consumption of large amounts of liquids with meals), this can allow the bacteria the opportunity to survive long enough to establish itself in the mucosal lining protected from stomach acid. This can lead to a very interesting paradox.
Currently, proton pump inhibitor drugs are your physician’s primary option for treating ulcers (along with antibiotics to kill the H. pylori) since they prevent your stomach from producing the stomach acid that is eating away at the exposed tissue. But without sufficient stomach acid, the bacteria can resist lower levels of stomach acid. That’s why the standard medical treatment for H. pylori requires antibiotics to kill the bacteria. The problem with this form of treatment, however, is that it may actually make the condition worse. What a quandary! In addition, when discussing H. pylori, it should be mentioned that only a small minority of people (5-10%) who have H. pylori in their system ever develop a peptic ulcer. Not just a quandary — but a paradox too!
The other two primary causes of peptic ulcers are non-steroidal inflammatory drugs (NSAIDS) and “social” drugs such as nicotine from smoking, alcohol, and caffeine. Many NSAIDS (especially aspirin) and corticosteroids irritate the stomach lining and can also cause ulcers. As for smoking, people who smoke are more likely to develop a peptic ulcer than people who do not smoke, and their ulcers heal more slowly. As for spicy foods and being stressed, they can make your ulcer “feel” worse, but there is no established link between them and the actual formation of peptic ulcers.
An alternative approach to ulcers
Supplemental digestive enzymes help digest so much of your meal during the 40-60 minutes of pre-digestion that your body requires a less sustained release of acid in the actual digestion phase. (Note: the strength of the stomach acid released is undiminished, only the time of exposure is reduced.) This means that taking digestive enzymes will lessen the amount of time that your stomach and duodenum are exposed to acid — but without raising pH when the acid is actually present. Those who suffer from chronic low levels of acid need not worry. Digestive enzyme supplements help here too by breaking down so much food in the pre-digestion phase that less acid is actually required overall. And over time, decreased demand results in increased reserve capability.
In addition, protease released with the stomach acid or present in the supplemental enzymes will begin breaking down the protective coating of the H. pylori bacteria. In other words, the protease will actually begin to digest the bacteria, rendering it vulnerable to stomach acid. However, for those with a severe existing ulcer, the protease may begin to digest damaged mucosal tissue because its protective coating is missing. This can cause noticeable discomfort for several days. To avoid this, when using digestive enzyme supplements, start with very small amounts of the supplement with your meals and build up slowly.
And then there’s mastic!
Mastic, which is widely used in Mediterranean cooking as a sweetening agent, offers a couple of interesting health benefits. First, studies now indicate that in addition to having direct antimicrobial activity, mastic renders H. pylori vulnerable to your body’s immune system. Mastic also enhances your body’s ability to regenerate the epithelial cells of your gastrointestinal lining. The net result is that mastic can help prevent and relieve a number of digestive disorders, including heartburn, gas, bloating, dyspepsia, nausea, and of course, peptic ulcers.
Acid reflux
 Acid reflux disease, also known as Gastroesophageal reflux disease or GERD, is defined as chronic symptoms or mucosal damage produced by the abnormal reflux of food and digestive juices (chyme) back up into the esophagus. This is commonly due to malfunctions in the lower esophageal sphincter that is supposed to prevent reflux from the stomach, back up into the esophagus — and to loss of control of acid production during the digestive process. Surprisingly, most treatments deal only with the second factor, not the first.
Acid reflux disease, also known as Gastroesophageal reflux disease or GERD, is defined as chronic symptoms or mucosal damage produced by the abnormal reflux of food and digestive juices (chyme) back up into the esophagus. This is commonly due to malfunctions in the lower esophageal sphincter that is supposed to prevent reflux from the stomach, back up into the esophagus — and to loss of control of acid production during the digestive process. Surprisingly, most treatments deal only with the second factor, not the first.
Before we can actually cover the causes of acid reflux and what you can do about it, we need to quickly review from the last newsletter, the phases of acid release, the regulating mechanisms that govern its release, and the triggers your body uses to signal for increased production of stomach acid. Understanding these triggers becomes the key to managing them and also exposes the flaws in the basic medical approach.
As we discussed last issue, there are three phases of stomach acid release. To quickly review:
Cephalic phase
Thirty percent of stomach acid is released by the anticipation of eating and the smell or taste of food. This is known as the cephalic phase, and as we will discuss in a bit, this is both governed and triggered by the vagus nerve. The vagus nerve starts in the medulla oblongata of the brain, runs down through the neck and then connects to virtually every organ in the body except the adrenal glands. As such it plays a major role in the digestive process — both sensing what’s happening in the stomach and signaling the stomach to prepare for the ingestion of food.
Gastric phase
Sixty percent of all stomach acid is released during the second phase of digestion, the gastric phase. This phase is triggered by the distention of the stomach — primarily the lower part of the stomach (the antrum) as chyme (the mixture of food and digestive juices) makes its way through the digestive process — and by the presence of proteins in the stomach. It is also triggered by a sudden rise in pH as stomach acid is diluted and if there is too little calcium in the blood. These four triggers cause gastrin, the primary regulator of stomach acid production, to be released. As we discussed in the last newsletter, gastrin is released into the bloodstream by the G cells located in the antrum of the stomach. Once in the bloodstream, gastrin circulates around body — ultimately reaching the cells of the stomach wall via the rich blood network that supports the stomach and bathes all of the cells in the stomach wall. Once there, gastrin works by stimulating the pariatel cells and the gastric chief cells to produce stomach acid and pepsinogen respectively as needed for digestion. In addition, gastrin causes the lower esophageal sphincter to constrict, thus inhibiting the backup of chyme and stomach acid into the esophagus. Disrupting this signaling mechanism causes the sphincter to relax, thus making it more prone to reflux.
Since these triggers for the release of stomach acid are so important, let’s review them in a little more detail.
- The first trigger is the anticipation of food — triggered either by the smell, sight, or imagining of food. This releases about 5-10% of the acid your stomach will produce for digestion. At this point, the esophageal sphincter is somewhat relaxed to allow food to more easily enter the stomach upon swallowing. However, since the acid content is so low and there is no food pressing up against the sphincter, this is not usually a problem phase when it comes to acid reflux.
- The next trigger is the distension of the stomach in the fundus and the main body of the stomach that happens when you eat your meal and food enters the stomach. As might be expected, the larger the meal, the greater the distension, and the stronger the signal telling the stomach to produce stomach acid. Two things are important to understand about this trigger. First, this is a weak trigger and is responsible for only about 5-20% of the acid your stomach produces. (In fact, only in abnormal circumstances does the total acidity in the stomach caused by the first two triggers combined climb much above 30% to 50% maximum. And second, as food passes out of the stomach and the distension lessens, the trigger also abates. An important point concerning acid reflux is that if you overeat, thus significantly stretching the stomach, you create huge backpressure on the esophageal sphincter — in effect, forcing food back up into the esophagus. The more you overeat, the greater the tendency to have acid reflux.
- Low acidity (high pH) while chyme is present in the stomach is the primary trigger for acid production in the stomach. In other words, if your body senses the presence of food that needs to be digested in the stomach and your stomach’s gastric mucosal chemoreceptors show too little acid to digest it, that will trigger the production of more stomach acid. Another way of looking at it is that the more you eat or drink (thus diluting your stomach juices), the higher the pH will climb and the more acid your stomach will be triggered to produce in order to lower that pH.
- Protein in particular is a trigger for acid production. As protein enters the stomach, it binds to hydrogen ions, thus neutralizing some of the acid and raising the pH of the stomach. As the pH rises, it lifts the inhibition of gastrin and HCl secretion. This triggers G cells to release gastrin, which in turn stimulates parietal cells to secrete more HCl. Low acidity in the stomach, whether the result of straight dilution or protein neutralization accounts for upwards of 60% of all stomach acid produced during digestion.
- Too little calcium in the blood (a condition called hypocalcemia) can trigger the production of additional stomach acid, beyond that required for ordinary digestion. It can certainly be caused by medical conditions such as deficient or ineffective parathyroid hormone (PTH), but I will leave discussion of those causes to your doctor. For our purposes, we are more concerned about the diet and lifestyle choices you make that might cause the condition and lead to the production of too much stomach acid. These include:
- Too much magnesium in the diet or through supplementation.
- Too little calcium in the diet.
- Too little vitamin D in the diet and/or too little exposure to sunlight. Vitamin D is required for calcium utilization by the body.
- Excessive use of magnesium based laxatives.
- The body going too alkaline, a condition called alkalosis. This can easily happen when people become obsessive about raising their body pH such as by drinking too much high pH water. Yes, having a slightly alkaline pH is essential for good health, and yes, the vast majority of people are too acidic because of high acid forming diets. But the body hates extremes, and it’s possible to become too alkaline, which can lead to hypocalcemia and too much acid in the stomach.
Intestinal phase
And finally, ten percent of stomach acid is released during the last phase of digestion, the intestinal phase. This is triggered when chyme begins leaving the stomach and causes distension of the duodenum. More importantly, though, the presence of chyme in the duodenum starts triggering the inhibition of gastrin release — and ultimately the inhibition of stomach acid production. This is regulated by the fact that the presence of chyme in the duodenum triggers the release of a number of hormones, including somatostatin, secretin, VIP, glucagon, calcitonin, and, of course, the appropriately named gastro inhibitory peptide.
Solutions to excess stomach acid
So now that we know the mechanisms that regulate the production of stomach acid and the triggers that lead to excess production in the stomach, we should be able to look at the alternatives for alleviating the condition — and what problems they might present.
Antacids
As we discussed last issue, once you understand the triggers involved in the production of stomach acid, you can instantly understand the problem with using antacids such as Tums and Rolaids. Although they effectively can neutralize excess stomach acid short term, the very act of raising pH in the stomach while food is present tells the body to produce more acid to compensate for the reduced acid levels. Thus, although you may get short term release from antacids, it is likely to be followed by another round of excess stomach acid.
 Drinking water
Drinking water
Drinking water to dilute excess stomach acid presents pretty much the same problem as using antacids. It will neutralize excess stomach acid short term, but by raising pH while the stomach is still distended, it will merely trigger the subsequent production of even more stomach acid.
Which brings up another issue associated with drinking water (or other liquids) while eating.
Drinking too much liquid while eating will dilute stomach juices from the get go. Not only does that interfere with digestion, it also immediately triggers the stomach to produce more stomach acid and is a primary factor in the onset of acid reflux disease. A little bit of water, wine, tea, whatever with your meal does not present a problem. Once you go beyond 8 ounces, however, problems start to develop. The more you drink, the greater the problems. Or to put it another way, three slices of pepperoni pizza sluiced down with an entire pitcher of root beer is a prescription for disaster.
Proton pump inhibitors
“Proton pump inhibitors” is the name of class of drugs that includes familiar names such as Nexium, Prilosec, and Prevacid. Right now, within the medical community — and within the public at large — proton pump inhibitors are among the hottest drugs in use. This is a testament both to the extent of digestive problems in the developed world and in the ability of these drugs to effectively stop production of excess stomach acid. How do they accomplish this miracle?
Without going into technical details, suffice it to say that proton pump inhibitors act by blocking an enzyme system that controls the final stage of the release of stomach acid from the parietal cells. Block the enzyme system, and you stop the release of stomach acid. How effective are proton pump inhibitors in stopping the release of stomach acid?
Quite simply, proton pump inhibitors can reduce gastric acid secretion by up to 99%.
Problem solved! If you had acid reflux before, you do not now. Even if some chyme is still backing up into the esophagus, it’s not a problem since there’s no stomach acid present. For doctors, it’s the perfect solution. It works like a charm, and their patients are happy.
However, since it doesn’t address the actual problem behind acid reflux and merely suppresses a symptom (which is in fact what most drugs do), it should not be surprising that there is a physical cost to regular use of these drugs.
But even more significantly, there is a fundamental problem with suppressing the production of stomach acid. Hydrochloric acid is not just “something” in the stomach; it is an essential component of the digestive process. Suppressing the symptoms of acid reflux by eliminating 99% of all stomach acid production presents a fundamental disruption of the digestive process. As you may remember from the last newsletter, hydrochloric acid is required for the digestion of proteins; it unwinds them so that pepsin can break them down. It is also required for the absorption of nutrients, particularly of vitamin B12. And it is required for the utilization and absorption of minerals such as calcium. Specifically, suppressing the production of stomach acid through the long term use of proton pump inhibitor drugs will lead to:
Incomplete digestion
Stomach acid denatures (unfolds) proteins so that they can actually be broken down by pepsin during digestion. Without being unfolded, they resist digestion. Without sufficient stomach acid present, this process won’t happen and the digestion of your food — particularly proteins — will be incomplete. This can result in long term deficiencies. In addition, since proteins now enter the intestinal tract not fully digested, this puts incredible stress on your pancreas (the digestive organ last resort, as it were) to produce vast quantities of protein digesting enzymes to try and compensate. And of course, the incomplete digestion of complex proteins (particularly those found in wheat, corn, and dairy) is a major factor in the onset of food allergies.
In addition, HCL kills many micro-organisms, such as the ones that travel into the digestive tract from the human mouth or come breeding in the food itself — as with contaminated meat or produce. Without sufficient stomach acid present, you are that much more likely to succumb to food poisoning and stomach flus — not to mention H. pylori and peptic ulcers, as we discussed earlier.
It is the presence of HCL in both the stomach and the duodenum that stimulates the flow of hormones, bile juices, and pancreatic juices in preparation for release into the small intestine. The less HCL produced, the less pancreatic juices are signaled for. Combine low HCL with no digestive enzymes being consumed with your food, and you have guaranteed lack of proper digestion (not just for proteins, but for fats too since the trigger for the release of bile has been disrupted).
Poor B12 absorption
Intrinsic factor is a protein made by the parietal cells in the stomach. It is made and released concurrently as the parietal cells make and release stomach acid. Effectively, the same things that trigger the release of stomach acid trigger the release of intrinsic factor — and more to the point, the same things that inhibit the release of stomach acid, such as proton pump inhibitors, inhibit the release of intrinsic factor.
Why is this important?
Because intrinsic factor is essential if your body is to absorb and utilize vitamin B12. The mechanism is simple. Intrinsic factor, if it’s present, binds with vitamin B12 in your food and/or supplements. This happens in the duodenum, and it accomplishes two things:
- First, it protects the B12 from bacteria that line the intestinal tract and that would “consume” it before your body could utilize it as it makes its way down to the ileum (the final section of the small intestine) where vitamin B12 is actually absorbed.
- Second, it plays a key role as part of an exchange mechanism that takes place in your ileum. In the ileum, the intrinsic factor bound to the B12 is swapped out for another protein, transcobalamin II (which is produced by epithelial cells that line the ileum). And it is this new complex that can pass through the walls of your intestine and travel to your liver, which ultimately regulates the utilization of B12 in your body.
Without intrinsic factor, most B12 could never reach the ileum, and even that which made it there could not be swapped out with transcobalamin II and thus utilized by the body. This means that if there is an intrinsic factor shortage, you will suffer from a B12 shortage, no matter how much you supplement.
The primary symptom of B12 shortage is pernicious anemia, a decrease in red blood cells that occurs when the body cannot properly absorb vitamin B12 from the gastrointestinal tract.
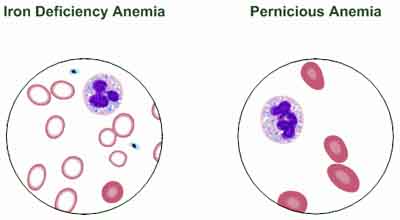
Symptoms of anemia can include:
- In people with anemia, the heart has to work harder to pump blood to get enough oxygen to the body’s organs and tissues. This stress on the heart can cause heart murmurs (an extra or unusual sound heard during the heartbeat), fast or irregular heartbeats, an enlarged heart, or even heart failure. Related symptoms include:
- Tiredness
- Paleness
- Dizziness when changing to standing position
- Rapid heart rate
- A lack of vitamin B12 can cause extra problems for the heart because it raises homocysteine levels. High levels of homocysteine add to the buildup of fatty deposits in blood vessels, which in turn can lead to heart attacks and strokes.
- A lack of vitamin B12 can damage nerve cells and cause problems such as tingling and numbness in hands and feet and problems with walking and balance. A vitamin B12 deficiency can cause changes in taste, smell, vision, and ringing in the ears. Finally, it can cause mental changes, including memory loss, confusion, and depression.
- Digestive tract. A lack of vitamin B12 may change the surface of the tongue and shrink or thin the stomach lining. Any changes that occur in the stomach can put a person at risk for stomach cancer. Related symptoms include:
- Stinging sensation on the tongue or smooth red tongue
- Cracked lips
- Yellow skin
Not surprisingly, pernicious anemia is a known side effect of the long term usage of proton pump inhibitors. And in fact, the problem is even worse than described above. In addition to causing B12 shortages, long term use of proton pump inhibitors leads to iron deficiency, which further exacerbates the problem by causing iron deficiency anemia — as we will now discuss.
Poor mineral absorption
Hydrochloric acid is essential for separating minerals from the foods that bind them. Or, if the minerals are already separated, as in supplements, low HCL levels in the stomach allow the minerals to recombine with the chyme into compounds that are difficult to absorb. Some minerals are more prone to this problem than others. Of the major minerals, iron, zinc, and calcium absorption in particular are directly affected by low acid levels.
In addition, if the stomach produces too little stomach acid, minerals such as calcium remain insoluble and cannot be ionized, which is necessary for assimilation in the intestines. Ionization is the process whereby an atom changes its structure so that it can combine with other elements. This is why chelated calcium, like many other chelates, is much more absorbable than raw calcium. The bottom line is that proper stomach acid levels are essential for ionic bonding which is necessary for intestinal uptake. The proper level of hydrochloric acid in the stomach is so important that its lack in the digestive process can account for as much as an 80% loss of available calcium absorption.
That means that regular users of proton pump inhibitor drugs are prone to be deficient in these minerals. In addition, sufficient stomach acid is essential for the absorption of most trace minerals. And considering that most people get almost none of these essential micronutrients in their diets to begin with, deficiencies of trace minerals is epidemic among people who suppress stomach acid production.
There’s more
In addition to the problems we’ve already discussed relative to B12 and minerals, the symptoms of HCL deficiency include:
- Bloating, belching, and flatulence immediately after meals
- Indigestion, diarrhea, or constipation
- Food allergies
- Candida overgrowth
- Acne
- Weak, peeling and cracked fingernails.
- And, surprisingly, heartburn
The bottom line is that despite the fact that proton pump inhibitor drugs can help eliminate the short term “symptoms” of acid reflux disease, they create a whole range of problems of their own associated with reduced stomach acid production and should not be used long term.
One final note on low stomach acid is that this is not just a concern for people who use antacids or proton pump inhibitor drugs. It is a major problem for the elderly. After a lifetime of eating enzyme deficient foods and forcing the stomach to overcompensate with extra high acid production, eventually the body’s capacity to produce stomach acid breaks down. At that point, no matter what you do, the body can no longer produce enough stomach acid to properly digest foods or negotiate the absorption of vitamin B12 and minerals. That’s one of the major reasons that so many of the elderly suffer from low blood counts and nutritional deficiencies — particularly mineral deficiencies.
Natural Health Alternatives
Fortunately, proton pump inhibitor drugs are not the only solution to acid reflux disease. There are natural alternatives. These include:
- Supplementing with digestive enzymes to reduce the need for stomach acid — thereby giving the body a chance to rest and recover its ability to produce sufficient stomach acid.
- Mixing one teaspoon of apple cider vinegar with water and a little honey and drinking this with each meal. You may gradually increase the vinegar up to 3-4 tablespoons in water if needed.
- And for the elderly who no longer produce enough stomach acid, supplementing with betaine hydrochloride (HCL) tablets can help, but anything beyond minimal doses as found in most health food store supplements should only be administered under the supervision of a health practitioner to avoid damage to the stomach lining.
For more on stomach acid and digestion, check out my 2007 newsletter on the subject.
And while discussing acid reflux disease, it’s important not to forget the physical contributors to the problem
- Hiatal hernia
- Poorly functioning lower esophageal sphincter
As we discussed in our overview of the digestive system, there are steps you can take to help alleviate hiatal hernias.
- Self massage
- Chiropractic adjustment
- Then, once you’ve corrected the initial hiatal hernia you might want to do some yoga exercises to strengthen your diaphragm so that your stomach won’t slip back up through the opening again. For example:
As for the esophageal sphincter, getting the release of stomach acid back into proper alignment and timing, can go a long way to helping the sphincter close properly — as can avoiding overeating.
Satiety/Weight Gain
 The human stomach can stretch quite a bit to accommodate a large meal, but also tends to collapse quickly after being stretched, which can cause hunger to return quite soon after a large meal. How much can it stretch? An unbelievable amount!! After a Thanksgiving dinner, it can stretch almost down to your pelvis. Then it empties — eventually — and you feel hungry again.
The human stomach can stretch quite a bit to accommodate a large meal, but also tends to collapse quickly after being stretched, which can cause hunger to return quite soon after a large meal. How much can it stretch? An unbelievable amount!! After a Thanksgiving dinner, it can stretch almost down to your pelvis. Then it empties — eventually — and you feel hungry again.
Grazing (eating a number of small meals throughout the day) on the other hand, does not overstretch the stomach, and keeps some food in there most of the day, which means you are constantly sending satiety signals to the brain. In other words, 6 small snack/meals will keep you feeling more satiated than 3 large meals — or any large meals, for that matter.
When it comes to stomach stretching, I always remember the possibly apocryphal stories of Diamond Jim Brady, the American businessman and financier of the later 1800’s, whose eating bouts were legendary.
No doubt, his appetite for gourmet food was insatiable, and he gorged himself at restaurants and parties. And as legend would have it, a typical Brady breakfast would include: eggs, pancakes, pork chops, cornbread, fried potatoes, hominy, muffins, and a beefsteak. For refreshment, a gallon of orange juice — or “golden nectar”, as he called his favorite drink. Lunch might be two lobsters, deviled crabs, clams, oysters and beef, with a few pies for dessert. The usual evening meal began with an appetizer of two or three dozen oysters, six crabs, and a few servings of green turtle soup, followed by a main course of two whole ducks, six or seven lobsters, a sirloin steak, two servings of terrapin and a host of vegetables. For dessert, he enjoyed pastries and a two pound box of candy.
Apocryphal or not, his lifestyle eventually caught up with him. Brady first consulted doctors for stomach diseases brought on by his uncontrollable eating habits: diabetes, heart and urinary problems, and high blood pressure. His prostate was swollen beyond belief. And his stomach was six times the size of a normal person’s stomach. After treatment at Johns Hopkins in Baltimore helped clean the prostate,Brady went back to New York and lived lavishly for another five years.
But on April 13, 1917, Brady died of a heart attack resulting from complications of his diseases. He left most of his wealth to Johns Hopkins and New York Hospital to help found medical institutes in his name.
Good health and good appetite!
Conclusion
And here we conclude our discussion of the stomach.
When next we continue with our series on the digestive system, we will pick up with a discussion of those organs just outside the alimentary canal that play key roles in the digestive process, including the:
- Pancreas
- Liver
- Gallbladder
In some ways, these are three of the most fascinating organs in the body — and three organs that are highly amenable to improvement through detoxing and flushing. Doctors absolutely do not understand the concept of detoxing when it comes to these organs, but we will explore the detox protocol using medical terminology and points of reference so that it will finally be understandable to them — as well as to you.
Continue reading the rest of Jon’s series about digestive health:
Overview of the Digestive System
Your Stomach, Part 1
Your Stomach, Part 2











I was advised to wean off of
I was advised to wean off of Prilosec OTC slowly to hopefully avoid
some of the rebound effect. It is ok to take d-linonene while still taking the Prilosec, or should I wait until I am completely off of the PPI?
Thank you.
This is without doubt the
This is without doubt the best article about heartburn and gastric problems i’ve ever encountered .
I suffer from heavy heartburn since over 6 years, i’ve visited “thousands” of doctors, and yet not one of them could provide me with even one quarter of the information that i’ve learned here.The only “treatment” that i got were tons of every single proton pump inhibitor that is avaible out there. Beside the several gastrcopies showing that these pills weren’t helping me at all (in fact it worsened constantly, from an simple antrum gastritis to a “highly active Pangastritis and GERD”), they increased the dosage from month to month, until i swallowed up to 100mg omeprazole daily with an age of 23. These pills gave me horrible sideeffects including every single as mentioned above + bloating, weight loss, irregular heartrate, panic attacks, sleeplessness, circulatory problems, depression, bad skin, muscle pain, really heavy chest pain and countless others. No one could tell me where these problems are coming from, they told me it was all “psychic”.Strangely, all those “psychical” conditions dissappeared after i decided to stop taking these horrible pills on my own after almost two years. The heartburn after stopping them was by far worse than before.
I’ve had a hiatal operation, but that also did not help. The acid has begun to attack my teeth and i really don’t know what to do anymore.
Anyway, long story, short message:
I’d like to thank you very very much for this excellent article, it provides a lot of useful knowledge and gives some hope that there is still a chance for healing this condition.
Thank you very much!
Sincerly, Igor from Germany.
Thank you for this article! I
Thank you for this article! I had H. Pylori two years ago and have been on PPI since. Thought I was getting better but now it’s back. In fact, I was sent for an abdominal US and they found some polyps in my gallbladder. The doc recommended I get my gallbladder taken out! I’m going for a 2nd opinion obviously but having a better understanding of what happens with the digestive system helps. I can have a more intelligent conversation with the specialist when I go in. I’m also going to try the alternative solution and see how it goes.
DITTO on the “best so far I
DITTO on the “best so far I’ve read on this topic” -and thanks for helping me approach my next meal and appointment with naturopath more intelligently so I don’t waste time and money. With my husband’s health issues and mine it has been quite stressful which I’ve always known needs to be relieved, too. Peace of mind to return my unopened Gaviscon and try again differently. An answer to prayer for guidance to my confusion and partial ignorance even after much research. Blessings,
Hi Baseline,
Hi Baseline,
In this article Jon mentions the problems with using antacids (it actually causes more stomach acid to be released). Can you tell me if apple cider vinegar with some bicarbonate of soda would have the same effect?
Sincerely, Tom
Note: Jon is specifically
Note: Jon is specifically referring to using antacids with food in the stomach. That said, apple cider vinegar would not be a problem, but adding bicarbonate of soda would be.
interesting, however 2
interesting, however 2 gastroenterologists have told me that because i was diagnosed with short non-dysplasia Barrotts Esophagus, I MUST take Omeprazole for the rest of my life, what would you say about that??
For obvious legal reasons, we
For obvious legal reasons, we cannot diagnose or prescribe for specific medical conditions—merely provide information. That said, this sounds like a trick question. if you were able to eliminate your acid reflux through other means, then why would you need to, or want to, continue taking proton pump inhibitors? Why would a responsible gastroenterologist continue to prescribe it? On the other hand, if you make no lifestyle changes, and do nothing to change your underlying condition so that your predilection for acid reflux remains undiminished, then yes, you will want to stay on Omeprazole for the rest of your life.
This is the best article
This is the best article about digestive problems I ever went through. I am researching every day for hours and it popped out first time.
This problem is new to me, about 3 weeks, and I am already off pills. My symptoms were little reflux with no heartburn and after 5 days on PPI I developed LPR symptoms. After days of search on internet (doctor looked at me like I am speaking Swahili when told I have heartburn first time of my life after using PPI), information about LOW acid instead of high seemed to me logical, but I didn’t understand the whole process in body. This article explained it amazingly. I am already on raw apple cider vinegar, but it’s just one day. People all around the world cannot say enough good things about this remedy.
Thanks again for doing this!
From last 8-9 months I am
From last 8-9 months I am suffering from low acid symptoms like fullness after meals, burping, constipation, low Iron, dry skin, acidity. I am currently taking digestive bitters from last two months so my symptoms have very less but if I stop taking them, all the symptoms reappears. Please advice to whom to consult and what to do?
I am allergic to yeast so I
I am allergic to yeast so I don’t take raw vinegar. Does filtered pasteurized apple cider vinegar work to supplement stomach acid?
Does lemon juice do the same?
You can drink a small amount
You can drink a small amount of fresh lemon juice or apple cider vinegar added to room temperature water about 15-20 minutes prior to eating to increase stomach acid. It is important that the water is not too cold because cold water can interfere with the digestion. If using lemon, squeeze half a lemon in 8 oz. of water. For apple cider vinegar, add 1 tablespoon in 8 oz. of water. If consuming apple cider vinegar or lemon juice in water regularly, I recommend drinking it through a straw and brushing your teeth well after in order to protect the enamel of your teeth.
Or you can just use a betaine hydrochloride supplement.
Hi, Jon Barron is against
Hi, Jon Barron is the best of the best. I suffered of artritis, gerd, gastritis, malnutrición,bowel issues,crónic fatigue, idiopatic muscle and joint pain, and you name mental disorders. First I discovered that I digested proteins, carbohydrates and fats is the culprit to many if not the main problems humans have at the moment.
Luck of hydroclloric acid is just terrible, can not absorve proteins, vitamins and clean the food of pathogens that come in the food chain. Pathogens then ferment the chime in the stomach, h pylori comes in destroying stomach membranes thus aggravating the hcl production. Pathogens also disrupt the liver,bile and gallbladder synergy and you end up with undigested carbohydrates and fats. Then you go to a MD that prescribe anti acids to further suppress hcl supply.
Luck of proteins is just death in advance, no tissue regeneration including bones,muscles, tendons cartilages, skin, hair, nails and you name it. Undigested carbohydrates is just like trying to run a car with no gasoline. Essential sugars for all membranes are lost and the damage is inmense. Your brain normal functions are disrupted because glucose is just not there. When fat is not properly metabolized, your brain not only malfunction for lunch of glucose but also starts to degenerate as well as cell membranes and hormones production is impaired.
Improper digestion also promotes the migration of beneficial bacteria from the large to the small intestine for undigested food, and the nightmare is vivid.
First, get your full digestion going with enzymes, attack pathogens and h pylori with an extra help of apple cider vinegar.
Then when you feel like your brain fog in going away, go for reasonable amounts of SATURATED, never PUFAs, along with the help of some bitters to produce better cholesterol in the liver to produce a powerful bile.
These simple steps will get your nutrition in balance. Only now start on probiotics to reduce even better eliminate your small intestinal bowel overgrow, but DO NOT ever take probiotics UNLESS you feed them right. Aim for fiber 30/40 hrs of fiber in fruits and vegetables NOT from grains, that will put you back in shape. IFFFFF possible eliminate grains in any form, whole or refined, they are NOT suitable for our guts and cross hybrids and GMOs only cause destruction and pain.
As for glucose fruits and veggies are the most reliable and healthy glucose supply there is.
Stay away from all sodas and processed foods, they are toxic and poison.
Research has shown that most people with SIBO that has been treated with antibiotics and diet eventually relapse with in short time, and guess what……it is because they have not taken digestive issues into account……
Good digestion and nutrition is where health comes back…….
Best wishes for you all…
Tito
As explained by Jon Barron,
As explained by Jon Barron, people who have weak immune systems; or take some medicines like Prednisone, etc., should not take any PROBIOTICS,since they contain acidophilus, causing inflammation of the heart lining. My question is:
For the applicable people here; what can they take in place of the PROBIOTICS? Also, are there any PROBIOTICS on the market that DO NOT contain a Lactobacillus Acidophilus ingredient?
Dr. Robert Bolmarcich
PH.D M.E.
I have acute hpylori. I have
I have acute hpylori. I have had Symptoms such as burning sensation in my stomach and surges of heat that overcomes my body, causing high temperature and Perfuse sweating. I have embarrassingly Bad breath, my stomach is constantly making noises as if there are aliens resident in my stomach. I recently started feeling discomfort on my right side behind the lowest part of my rib as if there is something protruding/ possibly a swelling. I am very scared, I definitely feel as though an ulcer is developing or worse. I was given antibiotics but refuse to take them as I know it will create other problems for me, plus, from what I have researched it eventually comes back. I would much prefer to go down the alternative root but do not know where to start.
You mentioned the need to maintain minimum acid formation using digestive enzymes and AVC along with mastic gum. How much of each should I take based on a worse case scenario? Is there anything else I should be taking. How do I begin to heal my gut. Would fasting help?
For obvious legal reasons, we
For obvious legal reasons, we can not diagnose or prescribe. But, I would search our site for your issue to read the articles we have on it. Plus, be sure to see our full series on the stomach:
Overview of the Digestive System
Your Stomach, Part 1
Your Stomach, Part 2