I devoted a full chapter in Lessons from the Miracle Doctors to an exploration of the immune system from an alternative health perspective, but today I want to start a series of newsletters that will expand on that work and fill in some of the missing details. To be sure, it would be easy to devote an entire book to the immune system — both because of its complexity and its importance — but for now, a series of newsletters will have to do.
Your immune system plays two vital roles in your body. First of all, it responds to foreign organisms that gain access to your body by producing antibodies and stimulating specialized cells that destroy the organisms or neutralize their toxic byproducts. In this manner, it defends against foreign invaders, including: germs, viruses, bacteria, fungi, and parasites. But it also stands guard over the cells of your body to ensure that they do not become abnormal or degenerate. Normally, your body produces anywhere from 100 to 10,000 abnormal cells a day in your body — produced as part of the normal metabolic processes, or as the result of exposure to environmental toxins or nutritional deficiencies. But if your immune system is functioning properly, it can identify each and every one of those cells and eliminate them before they can do any harm.
All in all, understanding how your immune system works and how to optimize it, while at the same time keeping it in balance so it does not run out of control, turn on you, and attack your own body, is essential to good health…if not your very life.
Your immune system
In many ways, your immune system is the most awesome system in your body, easily rivaling the brain in terms of complexity, subtlety, and “self-awareness.” As I mentioned a moment ago, your immune system is capable of identifying every single cell in your body and recognizing those that are friendly and belonging to your “self.” Conversely, it is also capable of singling out and identifying every single foreign invader that manages to make its way into your body — ranging from bacteria and viruses to fungi and parasites — and treating them as “non-self.” (Later on we will explore exactly how it accomplishes this remarkable feat.)
Once it has identified an invader, your immune system then quickly develops a customized series of defensive weapons that specifically target the invader’s weak link. It then begins building cellular factories that produce these weapons en masse, in quantities sufficient to totally overwhelm and crush the invader. Then, once the invader has been defeated, the immune system has the awareness to “shut itself down” until needed at a later date.
As amazing as all of this sounds, we haven’t yet come to the three most awe inspiring aspects of the immune system — the ones that highlight its intelligence. First, once it has defeated an invader, your immune system has the ability to “remember” that invader and the defense that was used to defeat it. If that invader ever makes another appearance, even years later, your immune system can launch its defense instantly — and at a level 1,000 times stronger than when the invader first appeared. In addition, your immune system can identify when a cell in your body has changed, has “gone over to the enemy” as it were — when it has turned from “self” into “non-self.” This is a stunning level of sophistication. Out of the trillions of cells in your body, your immune system can tell when a single one has mutated and become cancerous — and, in many cases, move in and destroy it before it can do any harm. In fact, it does this thousands of times a day, without missing a single mutant cell…we hope.
But most amazing of all, your immune system is in total communication with each and every part of itself. “So, what’s the big deal,” you might ask? “The brain does the same thing.” Yes, but remember, unlike the brain, the trillion+ cells of the immune system are not in physical contact with each other. To paraphrase Albert Einstein, “At its core, the immune system resembles nothing so much as a great thought.” And for good measure, your immune system is also in constant contact with your brain, not to mention every single cell in your body — not only receiving information from those cells, but also sending information and commands back to them so as to marshal systems and organs outside itself as required.
To put it simply, your immune system is awe inspiring!
The anatomy of the immune system
 Before we even begin to look at the immune system, we need to define what we’re talking about when we refer to the immune system, which is not quite as simple as it might first seem and is actually open to some debate. Generally speaking, though, the body’s immune system is comprised of those cells and organs that contribute to the body’s active response against foreign invaders and self-generated rogue cells — “active” being the operative word. The immune system, then, excludes non-specific defenses — primarily physical barriers — such as the skin, respiratory tract, and lining of the intestine. The inside of the nose, for example, is lined with mucous to capture pollen and dust and prevent them from entering the body before they can become a problem. That is an immune function, but since it is both passive and non-specific, it’s not counted as part of the immune system. But what about the 100 trillion beneficial bacteria that line every square inch of a healthy intestinal tract, from your mouth to your anus? They not only provide a passive barrier to invading pathogens, but a number of them actively kill those same pathogens. Others produce immune-boosting biochemicals such as transfer factor and lactoferrin. L. plantarum, for example, has the ability to eliminate thousands of species of pathogenic bacteria. That’s a pretty “active” immune function. And then there’s acidophilus. It can produce a number of powerful antimicrobial compounds in the gut (including acidolin,1 Hamdan IY, Mikolajcik EM. “Acidolin: an antibiotic produced by Lactobacillus acidophilus.” J Antibiot (Tokyo). 1974 Aug;27(8):631-6. <http://www.ncbi.nlm.nih.gov/pubmed/4373425> and bacteriocins such as acidophilin and lactocidin.2 Luc De Vuyst, Frédéric Leroy. “Bacteriocins from Lactic Acid Bacteria: Production, Purification, and Food Applications.” J Mol Microbiol Biotechnol 2007;13:194–199 <http://mywebspace.quinnipiac.edu/erdonnald/BI298/Bacteriocins.pdf> If present, beneficial bacteria in your intestinal tract can account for 60-70% of your immune system’s activity. Nevertheless, in accordance with standard medical texts, for the purposes of these newsletters, we will consider them outside our definition of the core immune system proper.
Before we even begin to look at the immune system, we need to define what we’re talking about when we refer to the immune system, which is not quite as simple as it might first seem and is actually open to some debate. Generally speaking, though, the body’s immune system is comprised of those cells and organs that contribute to the body’s active response against foreign invaders and self-generated rogue cells — “active” being the operative word. The immune system, then, excludes non-specific defenses — primarily physical barriers — such as the skin, respiratory tract, and lining of the intestine. The inside of the nose, for example, is lined with mucous to capture pollen and dust and prevent them from entering the body before they can become a problem. That is an immune function, but since it is both passive and non-specific, it’s not counted as part of the immune system. But what about the 100 trillion beneficial bacteria that line every square inch of a healthy intestinal tract, from your mouth to your anus? They not only provide a passive barrier to invading pathogens, but a number of them actively kill those same pathogens. Others produce immune-boosting biochemicals such as transfer factor and lactoferrin. L. plantarum, for example, has the ability to eliminate thousands of species of pathogenic bacteria. That’s a pretty “active” immune function. And then there’s acidophilus. It can produce a number of powerful antimicrobial compounds in the gut (including acidolin,1 Hamdan IY, Mikolajcik EM. “Acidolin: an antibiotic produced by Lactobacillus acidophilus.” J Antibiot (Tokyo). 1974 Aug;27(8):631-6. <http://www.ncbi.nlm.nih.gov/pubmed/4373425> and bacteriocins such as acidophilin and lactocidin.2 Luc De Vuyst, Frédéric Leroy. “Bacteriocins from Lactic Acid Bacteria: Production, Purification, and Food Applications.” J Mol Microbiol Biotechnol 2007;13:194–199 <http://mywebspace.quinnipiac.edu/erdonnald/BI298/Bacteriocins.pdf> If present, beneficial bacteria in your intestinal tract can account for 60-70% of your immune system’s activity. Nevertheless, in accordance with standard medical texts, for the purposes of these newsletters, we will consider them outside our definition of the core immune system proper.
However, that still leaves the Complementary Immune System, comprised of a series of cascading enzymes, which although many medical texts do not include, we will…and we’ll discuss it later. For now, though, let’s focus on those parts of the immune system that are specific, wide-ranging, and have long term memory, which is the standard medical definition of active immunity. In addition, as we discussed earlier, the active immune system can distinguish “self” from “non-self” with great accuracy and subtlety. An example of its subtlety is that despite its tendency to reject anything that is foreign, it is, nevertheless, extremely tolerant of foreign substances that it encounters during embryologic development and early infancy. That means that if you expose a fetus to a foreign body during development, it will not reject it, but rather recognize it as “self” from that point forward. Why is this so crucial? Because it allows the mother to pass natural antibodies to the fetus across the placenta, or to the baby in breast milk, thus helping to protect the child with the mother’s immunity before the baby builds its own immune function, not to mention facilitating the actual building of the child’s immune system. This, of course, is crucial to the child’s survival.
The anatomy of the immune system
 All blood cells, both red and white, begin as stem cells in your bone marrow. These undifferentiated cells begin to assume individual characteristics and become either red cells (the oxygen carriers) or white cells (the cells of the immune system). Further differentiation divides the white cells (also called leukocytes) into four main types of cells:
All blood cells, both red and white, begin as stem cells in your bone marrow. These undifferentiated cells begin to assume individual characteristics and become either red cells (the oxygen carriers) or white cells (the cells of the immune system). Further differentiation divides the white cells (also called leukocytes) into four main types of cells:
- Lymphocytes
- T-cells
- Helper cells
- Natural killer (NK) cytotoxic T-cells
- Suppressor T-cells
- Memory T-cells
- B-cells
- Phagocytes
- Granulocytes
- Dendritic cells.
Let’s explore them in more detail.
Lymphocytes
Lymphocytes are white blood cells that detect and destroy foreign invaders in various ways. They serve as the key operatives of the immune system. In a healthy body, not under attack, they number about one trillion. There are three main classes of lymphocytes: T-cells, B cells and natural killer (NK) cells. Under a microscope, T and B cells are indistinguishable from each other. It wasn’t until specific antibody testing procedures were developed that scientists were able to differentiate between them. NK cells are easily spotted because they are noticeably larger than either T or B cells.
T-cells
T-cells work primarily through what is known as cell mediated immunity, which is another way of saying that they do not instantly attack invaders on their own authority like antibodies do, but rather require other cells to activate/mediate their response. In addition, they work not so much by “directly” attacking invaders but by attacking cells that have been infected by invaders and inside which the invaders are hiding. In other words, T-cells primarily defend against viruses that have taken up residence in host cells and cells that have turned malignant. This is in contrast to B-cells and antibodies that take on invaders that work out in the open in your bodily fluids — most notably bacteria.
Another difference between T-cells and other cells of the immune system is that T-cells are smarter than the other guys. They’ve been to school, as it were.
When T-cells leave the bone marrow, they have no functioning TCRs (T-cell receptors) and are virtually identical to each other, and essentially non-functional. In fact, they are called pre-T-cells, or pre-TCR T-cells. But then they go to “school” in the thymus, which is where they migrate to after leaving the bone marrow. The thymus is a small fatty gland located under the collar bone. It “curls up” after childhood and is difficult to find in adults, but it’s still there. Incidentally, the “T” in T-cells refers to the fact that they mature in the thymus. While in the thymus, they learn two things. First, they are educated (programmed by certain thymic proteins) in how to distinguish between the cells of the body and invading cells, and how to distinguish between normal healthy cells and mutated rogue cells. This is where they learn to distinguish between “self” and “non-self.” T-cells that cannot make this distinction are eliminated so that they do not make their way into the body and begin attacking it.
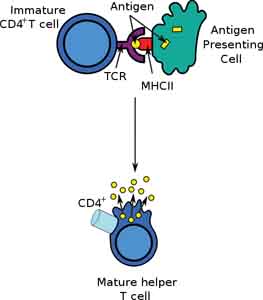 In addition, the pre-T-cells are also exposed to proteins associated with virtually every invader that the body has ever encountered in its existence –or that has been passed on to it by its mother. Like young children that have very malleable minds, the DNA associated with the non-functioning T-cell receptors is very malleable and can be easily rearranged in multiple variations, thereby creating millions of different T-cells with millions of different TCRs. TCRs, by the way, are protein molecules on the surface of T-cells that are responsible for recognizing specific protein molecules found on the surface of each different invading antigen. Each and every one of these “new” TCRs is now functional in that it can respond to each and every one of the invaders whose protein was used to train the T-cell. Those cells that fail this part of training (that have unstable TCRs) are eliminated. But those that survive differentiate into either CD4+ or CD8+ T-cells depending on which class of antigen they are trained to recognize. If their TCR recognizes an MHC class I antigen (as found on nucleated cells, such as cells infected with a virus), they become T8 cells. If their TCR recognizes an MHC class II antigen (as found on other immune cells such as macrophages, T8 cells, B cells and dendritic cells), they become T4 cells. This is important because MHC proteins act as “signposts” that serve to alert the immune system if “non-self” material, such as a virus, is present inside a cell. They achieve this by displaying fragmented pieces or antigens on the host cell’s surface.
In addition, the pre-T-cells are also exposed to proteins associated with virtually every invader that the body has ever encountered in its existence –or that has been passed on to it by its mother. Like young children that have very malleable minds, the DNA associated with the non-functioning T-cell receptors is very malleable and can be easily rearranged in multiple variations, thereby creating millions of different T-cells with millions of different TCRs. TCRs, by the way, are protein molecules on the surface of T-cells that are responsible for recognizing specific protein molecules found on the surface of each different invading antigen. Each and every one of these “new” TCRs is now functional in that it can respond to each and every one of the invaders whose protein was used to train the T-cell. Those cells that fail this part of training (that have unstable TCRs) are eliminated. But those that survive differentiate into either CD4+ or CD8+ T-cells depending on which class of antigen they are trained to recognize. If their TCR recognizes an MHC class I antigen (as found on nucleated cells, such as cells infected with a virus), they become T8 cells. If their TCR recognizes an MHC class II antigen (as found on other immune cells such as macrophages, T8 cells, B cells and dendritic cells), they become T4 cells. This is important because MHC proteins act as “signposts” that serve to alert the immune system if “non-self” material, such as a virus, is present inside a cell. They achieve this by displaying fragmented pieces or antigens on the host cell’s surface.
Once matured in the thymus, every T-cell carries a distinctive molecule on its surface that affects how it behaves. This is the T-cell receptor that we’ve already talked about. T-cell receptors are actually protein molecules that function as “locks” for the MHC “keys” found on the surface of antigens, and other cells of the immune system. (We will discuss this in more detail later.) Antigens, by the way, are foreign molecules or substances that provoke an immune response from the body by virtue of specific geometric arrangements (antigenic determinants) on their molecular surfaces. An antigen could be a grain of pollen, a speck of dust, a bacterium, a blood cell, or a simple molecule. The bottom line is that something on the molecular surface of the antigen is recognized by the immune system as “non-self.” So once again, the MHC molecule “keys” tell the immune system “locks” what’s inside the antigen — whether it’s friend or foe — according to what molecular lock on the T-cell’s surface the key fits into. It should be noted, however, if we wish to press our lock and key analogy a little further, that although TCRs are unique and number in the millions, we’re nevertheless talking about a low security skeleton key system here as opposed to a high security vault key. That is to say, despite the tendency to specificity, many different T-cell receptors recognize the same antigen, and many antigens are recognized by the same TCR.
The TCRs are named after the proteins they present on the surface of the T-cells on which they reside according to a naming convention referred to as the “cluster of differentiation” (CD) protocol. Thus a cell that carries a cluster of differentiation 4 protein on its surface is called a CD4+ T-cell (aka a T4 cell). If it carries an 8 protein, it is called a CD8+ cell (aka a T8 cell). Although there are a number of different CD cells (CD1, 2, 3, and 17, for example), the two primary types of T-cells are CD4+ and CD8+. T4 cells are known as helper T-cells because they don’t attack invaders themselves, but rather identify foreign invaders, activate B-cells, other T-cells, natural killer (NK) cells, and macrophages to attack the invader. T-8 cells, on the other hand, are known as cytotoxic T-cells (also called “killer cells”), which — once activated by T4 cells — attack and destroy rogue mutated cells in the body or cells that have been invaded by viruses and compromised. Because this is a two-step process that requires the T4 cells to mediate both the recognition of an invader and the subsequent attack on that invader by the killer cell, it is often referred to as cell-mediated immunity.
 Also, as I mentioned earlier, T-cells defend against intracellular pathogens — things that happen inside other cells or by other cells. Viruses, for example, are intracellular pathogens. They work inside cells. Until a virus implants itself inside the cells of your body, it can neither replicate nor do any harm. Fortunately, even though hidden inside your body’s own cells, the viruses leave traces of themselves on your cells’ surfaces, which usually allow your immune system to track the viruses down. Additionally, some cancers, which also use your own body’s cells to harm you, present protein markers on their cell surfaces, just like viruses, that allow your immune system to perceive them as “non-self” foreign bodies (much like virus infected cells) and thus marked for destruction…if your immune system is functioning properly.
Also, as I mentioned earlier, T-cells defend against intracellular pathogens — things that happen inside other cells or by other cells. Viruses, for example, are intracellular pathogens. They work inside cells. Until a virus implants itself inside the cells of your body, it can neither replicate nor do any harm. Fortunately, even though hidden inside your body’s own cells, the viruses leave traces of themselves on your cells’ surfaces, which usually allow your immune system to track the viruses down. Additionally, some cancers, which also use your own body’s cells to harm you, present protein markers on their cell surfaces, just like viruses, that allow your immune system to perceive them as “non-self” foreign bodies (much like virus infected cells) and thus marked for destruction…if your immune system is functioning properly.
On a related note, tissue transplants (lung, heart, kidney, etc.) are also viewed as intracellular foreign bodies — unless coming from an identical twin — and thus subject to an all-out attack by the body’s T-cells. This can quickly lead to rejection of the transplanted organ. That’s why transplant recipients require immunosuppressants for the rest of their lives to prevent such an attack. That said, even if you use immunosuppressants, there will be a continual low level attack on the transplanted organ that over time can lead to the degradation of the transplanted organ. But immunosuppressants can push that date out many, many years.
It should be noted that we don’t store a lot of T-cells, just a few of each kind. Storage areas include the spleen, liver, and Peyer’s patches in the intestinal tract. The T-cells wait there, looking to see if they recognize the particular protein geometry of an invader they have been trained to identify and then react to it.
Helper cells
This is pretty much another name for T4 cells (CD4+). T4 cells are known as helper T-cells because they help activate the antibody-mediated immune response by identifying foreign invaders, then help activate B-cells, other T-cells, natural killer (NK) cells, and macrophages to attack the invader. In addition, T4 cells produce interlukin-2 to costimulate virtually all immune processes.
Cytotoxic NK (natural killer) T-cells
These are mainly T8 cells that have been activated by T4 cells and “transformed” into killer T-cells (AKA, Killer CD8+ T-cells). Remember that we mentioned earlier how the T-cell receptors on T4 cells are tuned to identify and locate other cells in the immune system in addition to “non-self” invaders? Well, NK T-cells are the reason why. T4 cells identify the invading threat and then hunt down T8 cells to transform them into killers to crush the invader. Once activated/transformed by the T4 cell, the T8 cell undergoes further growth and differentiation when stimulated by interleukin-2 released by the same T4 cell that locked onto it and activated it. This exponentially increases the number of NK cells programmed to identify a target antigen and then travel throughout the body in search of antigen-positive cells to destroy. This provides a failsafe two-step costimulation process for your immune system — stimulation by direct contact with the T4 cell and stimulation by the interleukin-2 released by the same T4 cell. This two step process helps prevent the immune system from misfiring and attacking healthy cells in the body. This is so important that it’s worth discussing in a little more detail.
A car provides a useful metaphor for thinking about the way costimulators (interleukins, AKA cytokines) work. If antigens on foreign invaders are the “keys” that turn on the T-cells’ engines, then costimulators, are like “putting the car into gear” after you turn the engine on. In the same way that you wouldn’t want a car to jump forward the moment you turn the ignition key, you wouldn’t want your immune system to jump into action until the nature of the invader was truly verified. In this sense, the interleukins serve as a failsafe backup mechanism.
The literal translation of interleukin is, “To speak between white cells.” Once it has been determined that the threat registered by the T-cells is legitimate, the interleukins will communicate between the T4 and T8 cells to throw the immune system into action by up-regulating its activity. A question you might have at this point is, “Just exactly what are costimulators protecting against?” In simple terms, the failsafe system is required to make sure the body doesn’t throw itself into an overly strong allergic reaction and possible anaphylactic shock. When the system doesn’t work properly, we have exaggerated allergic responses to seemingly innocuous situations. This can sometimes even result in death.
All of that said, after recognition of the invader and costimulation by interleukin, the T8 cells are sensitized and transformed into cytotoxic killer cells; after which, they then begin to clone themselves into millions and millions of identical killer cells with similar TCRs — all primed to attack the new invader.
Killer T-cells recognize specific “non-self” cells (primarily intracellular foreign invaders) and kill them by lysing (breaking apart) the compromised cells. They require direct contact to kill cells. Doctors call this the “kiss of death.” The reason for this is that the CD8 key of the activated Cytotoxic T-cell has to directly insert itself into the specific Antigen-MHC-1 TCR protein complex on the compromised cell that it is trained to recognize. Once this lock and key mechanism attaches, matches, and inserts, it sets up a series of responses.
- The Cytotoxic T-cell secretes a protein called perforin, which causes the invader’s plasma membrane to blow holes in itself, thus causing the cells’ contents to spew out and the infected cell to die. If the invader is a virus that has taken up residence within the cell, killing the cell prevents the virus from reproducing. In addition, macrophages eat up any viruses that flow out from the broken cell (see below).
- NK cells secrete lymphotoxin, which kills enzyme systems in the invader.
- They secrete gamma interferon, which stimulates macrophages to eat foreign invaders. Thus any viruses that were released from inside the cells are now fully exposed and subject to destruction by the macrophages. (As we mentioned earlier, viruses are intracellular foreign invaders. They can’t work, reproduce, or do damage by themselves. They are literally DNA surrounded by a protein coating. They have to take over another cell and its energy producing mechanisms in order to replicate and spread damage. Once a virus enters a human cell, in most cases, it is protected inside that cell. However, it will leave telltale traces of protein on the cell surface that will tell the immune system to no longer consider that cell as part of “self” and attack the cell in which it has taken up residence. Blowing apart that cell exposes the virus to the other elements of the immune system capable of destroying it. )
Once they have done their job killing a cell, cytotoxic (killer) T-cells can detach and find another invader — thus killing again, and again, multiple times, before they are used up and die.
Suppressor T-cells
So what happens when the invader is destroyed? How does the immune system know the battle has been won, and that it’s time to take a rest? That’s where Suppressor T-cells come into play. These cells cause down-regulation of the immune response. Very little is known about them. But medical researchers do know that when the immune response has gone on long enough and that the invader has been defeated, suppressor cells somehow signal the rest of the immune system to ratchet down so that it doesn’t start harming healthy cells. The bottom line is that we don’t yet know how they do these things — only that they do.
It should be noted that some cancer cells have the ability to convince suppressor T-cells to tell the rest of the immune system to shut down prematurely so that the immune system won’t attack the cancer cells — thus allowing the cancer to spread unopposed. Very devious. Very nasty.
Memory T-cells
Once an invader is defeated, most of the active T4 and T8 cells dry up and disappear. However, the T4 cells produce a clone of themselves called Memory Helper T-cells, which can last a long, long time (anywhere from decades up until the day you die) to resist the next invasion of that specific antigen. This is an amazing advantage when you consider that building that first recognition response can take up to a week or ten days. That’s a lot of time for an invader to have free rein in your body — more than enough time to make you extremely sick, or even kill you when faced with an aggressive invader. However, having Memory Helper T-cells in place cuts that time to just a matter of hours.
Consider the example of a mother taking care of her child with measles. How does her previously acquired immunity prevent her from getting measles? Surprise! It actually doesn’t. Every mother actually catches measles from her child no matter how many times she’s been vaccinated or had measles herself. But because of her immune system’s “memory” of measles, her immune system’s response is so fast that she totally eradicates the invader before she even gets one single symptom. This is a really, really important point to understand about your immune system. Even if you have immunity, you’re still going to get the disease, if the virus enters your body. The virus absolutely starts reproducing in your body, but the response is so fast that it kills the virus before it can ever really get started. It’s gone before you ever even knew it was there. That’s how immunity works. But it’s not only the speed of the response that’s increased, it’s also the strength. The response triggered by memory cells during second exposure to an invader can be as much as a 1000 times stronger than the initial response. This is known as an anamnestic (or accelerated) response. And that’s how a trained immune system protects you against a foreign invader — virus, bacteria, fungus, whatever. And in fact, approximately one in every 200 immune systems can even protect against seemingly unprotectable viruses such as HIV AIDS. Pretty amazing, not to mention being indicative of opportunities for optimizing your immune system through both natural and medical means.
Conclusion
That concludes our discussion of cell mediated immunity — T-cell immunity, mediated by lymphocytes, interferon, and interleukins. In Part 2 of our series on the Immune System, we’ll explore:
- Humoral immunity, which takes place in the blood. B cells, the primary agents of humoral immunity.
- Antibodies, the chief weapon of the humoral immune system.
- Phagocytes — the “Sin Eaters” of the immune system.
- Granulocytes. Think of them as bounty hunters or hired guns.
- Dendritic cells. These are kind of like super heroes with special powers. Spiderman comes to mind.
- The organs of the immune system.
- And how to boost your immune system.
In subsequent parts we’ll also explore some of the diseases of the immune system, how to rebalance your immune system, Circulating Immune Complexes, the Complementary Immune System, how to give your immune system a break and directly kill invading pathogens, how your immune system communicates with itself, the pros and cons of immunization, and how medical science is both helping and hurting the immune system.
Click below if you’ve missed any part of this Immunity series:
Anatomy and Physiology of the Immune System, Part 1
Anatomy and Physiology of the Immune System, Part 2
Anatomy and Physiology of the Immune System, Part 3
Anatomy and Physiology of the Immune System, Part 4
References
| ↑1 | Hamdan IY, Mikolajcik EM. “Acidolin: an antibiotic produced by Lactobacillus acidophilus.” J Antibiot (Tokyo). 1974 Aug;27(8):631-6. <http://www.ncbi.nlm.nih.gov/pubmed/4373425> |
|---|---|
| ↑2 | Luc De Vuyst, Frédéric Leroy. “Bacteriocins from Lactic Acid Bacteria: Production, Purification, and Food Applications.” J Mol Microbiol Biotechnol 2007;13:194–199 <http://mywebspace.quinnipiac.edu/erdonnald/BI298/Bacteriocins.pdf> |












Thank you. That was so well
Thank you. That was so well done. Looking forward to the next installment on the immune system.
Isn't this remarkable
Isn’t this remarkable information that’s so readily available to lay persons like me, that is life changing, and so helpful?
Thank you so much! This info
Thank you so much! This info was very informative and yet easier to understand. I,too, look forward to more info, specifically on how to the B cells.
Thanks for the refresher
Thanks for the refresher course, I’ve been away from this subject for quite a while, and this article brought a lot back.
This is the best explanation
This is the best explanation of the immune system I have ever read . Thank you
Sir,
This is an excellent
Sir,
This is an excellent information regarding immune system.We are living in a toxic world.Our immune system can only liberate us and helping us to live disease free life.Miracles are done by our immune system in doing so.Many researches are on the way and more and more investigations have been made .Send me more exiting medical wonders of this system.Thank you-yours-N.Vijayan from India
Hi Jon
What a really awesome
Hi Jon
What a really awesome system we have!
What an awesome article you’ve written!
Thanks for the continuing education and re-education. To me, what you’ve said has not only opened my mind to the immune system and its amazing capabilities, it has also opened my mind to deeper levels of myself that I’ve to learn to be aware of.
Would that I could learn from my immune system to differentiate between what’s truly ‘self’ (at all levels) and ‘non-self.’
Can’t wait to read the next part of your article.
John (from Singapore)
This information is so
This information is so crucial to my understanding of myself. Thanks a lot.
Hi Jon. While the above
Hi Jon. While the above article is very interesting to read, it reflects "old type" thinking of the medical establishment. (pathogenic) viruses are a figment of the conventional medical establishment and have never, to this day been proven to exist. What is commonly referred to as viruses, are bits of protein, made by the body cells for the body to use as "building materials" to repair damage caused either through trauma (injury) or by a specific biological SPECIAL program of Nature. The so called viruses found on the cervix are a case in point. Cervical 'cancer' is such a PROGRAM of Nature, which has two phases to it. Phase one is an ulceration caused by the perception of the Psyche, that fertilization did not, or could not take place, constituting a biological conflict. Upon resolution of this conflict, phase two is initiated and the ulceration is being restored to normalcy, using said 'virus material'.
Awesome work. I have some
Awesome work. I have some questions to which the answers may be very obvious and may require some thought.
Why is it that Cancer Attracts Gadolinium and Ferrous Oxide?
Why is it that academia has never seen a molecule?
Why is it that a 1″ Cube Neodymium Iron Boron Magnet Pick up Walmart Iron Pills.
Why did the attraction to said pills increase after 3 days?
Does an MRI which is 300 to 600 time stronger than said magnet rip iron through your cells?
Why does Cancer Glow in MRI, X-rays, Cat/Pet Scans?
Why does Cancer spread after so many tests?
Why does a teaspoon arc in a lesser radiation device called a microwave?
Why does a Blood Expert have his brain click into a new logical form when reading these questions?
IS CANCER = MAGNETS?
I KNOW what Cancer is, do you?